HKUMed Identifies Novel Host Protease Determinants for SARS-CoV-2 Infection
The study revealed additional protease determinants for the infection of ancestral SARS-CoV-2 and SARS-CoV-2 Omicron BA.1, which contribute to our understanding of the biology of coronavirus entry.
Researchers from Department of Microbiology, School of Clinical Medicine, LKS Faculty of Medicine, the University of Hong Kong (HKUMed), has identified novel host protease determinants, that facilitate the infection of SARS-CoV-2, including the Omicron variant, which provided new targets for combating the pandemic. In addition to the host protease determinants, members from the membrane-type matrix metalloproteinase (MT-MMP) and a disintegrin and metalloproteinase (ADAM) families were found to be able to mediate SARS-CoV-2 entry, with an increase efficiency against Omicron BA.1. This finding suggests that a new treatment strategy at MMP inhibition should be explored to effectively combat Omicron BA.1 and other Omicron sublineages. The research has now been published in the international scientific journal Science Advances [link to the publication].
Background
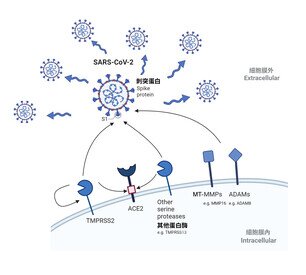
SARS-COV-2 is highly transmissible among people. In the past three years, more than 650 million cases were found in 200 countries or regions globally, recorded more than 6.68 million death cases.1 Infection of SARS-CoV-2 requires proteolytic cleavage of the viral spike protein with host proteases. Recent evidence suggested that in addition to TMPRSS2, other transmembrane serine proteases such as TMPRSS4, TMPRSS11D, and TMPRSS13 can similarly activate SARS-CoV-2 spike at the plasma membrane. However, the potential role of alternative transmembrane proteases in facilitating SARS-CoV-2 entry remains only partially understood. A comprehensive investigation of the host transmembrane protease determinants that contribute to efficient entry of SARS-CoV-2 is a key research question that can facilitate our understanding of the biology of SARS-CoV-2 infection and pathogenesis, and may provide new targets of intervention.
Research methods and findings
By using pseudovirus screening system, the team identified the involvement of membrane-type MT-MMP and ADAM in SARS-CoV-2 cell entry. The physiological importance of MT-MMP mediated SARS-CoV-2 cell entry was then evaluated using in vitro and in vivo model. Treatment of pan-MMP inhibitor reduced the amount of SARS-CoV-2 by 96% (p < 0.0001) and 85% (p <0.0001) in human lung and intestine cells, respectively. The researchers showed that inhibition of MT-MMPs significantly reduces SARS-CoV-2 replication, indicating MT-MMP inhibition can be explored as a new anti-SARS-CoV-2 strategy in the cell model.
In the hamster model, pan-MMP treatment significantly reduced the infectious viral load of SARS-CoV-2 (p < 0.0001) and alleviated the pathogenesis in the hamster lungs. More interestingly, the team found that SARS-CoV-2 Omicron BA.1 has an increased efficiency on MT-MMP usage in comparison to that of the ancestral SARS-CoV-2 and pan-MMP inhibitor demonstrated higher potency in inhibiting SARS-CoV-2 Omicron replication in the nasal turbinates and lungs of hamsters, compared to the serine protease inhibitor, camostat. Mechanistically, they showed that MT-MMPs can cleave SARS-CoV-2 spike (S) and a receptor protein called angiotensin-converting enzyme 2 (ACE2), and facilitate spike-mediated fusion. These findings revealed additional protease determinants for SARS-CoV-2 infection and contributes to our understanding on the biology of coronavirus entry.
Significance of the study
Dr Jasper Chan Fuk-woo, Clinical Associate Professor, Department of Microbiology, School of Clinical Medicine, HKUMed, commented, ‘The study revealed additional protease determinants for the infection of ancestral SARS-CoV-2 and SARS-CoV-2 Omicron BA.1, which contribute to our understanding of the biology of coronavirus entry. In addition, these findings are important for the design of new drug treatment against the current predominant SARS-CoV-2 variant, Omicron.’
About the research team
The research team is led by Dr Chu Hin, Assistant Professor, and Dr Jasper Chan Fuk-woo, Clinical Associate Professor, Department of Microbiology, School of Clinical Medicine, HKUMed.
https://www.hku.hk/press/news_detail_25842.html